Pancreatic ductal adenocarcinoma (PDAC) is the most common cancer of the pancreas. PDAC is an aggressive and difficult malignancy to treat. PDAC has increased in incidence by 0.3% annually since 2006 and is expected to become the second cause of cancer-related death in the United States in the year 2030.
Pancreatic ductal adenocarcinoma (PDAC) explained
PDAC is a type of exocrine pancreatic cancer that accounts for more than 90% of
pancreatic cancer cases. It develops from cells lining small tubes in the pancreas called ducts. These carry the digestive juices, which contain enzymes, into the main pancreatic duct and then on into the early part of the small bowel called the duodenum.
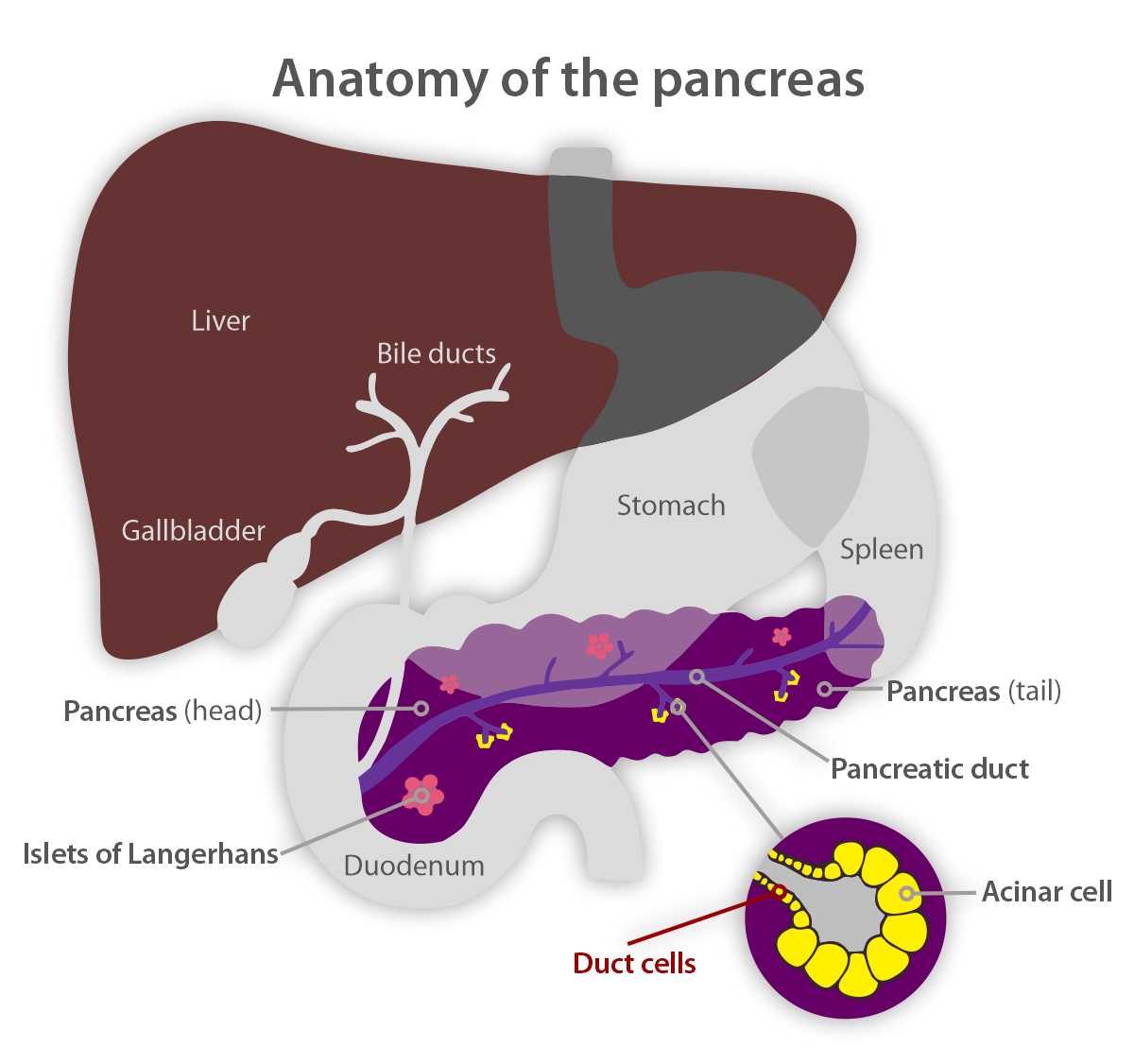
PDAC can grow anywhere in the pancreas, though it is most often found in the head of the pancreas. Symptoms can include abdominal and/or back pain, weight loss, changes in bowel habits, discoloration of eyes, skin, urine, or stool, or even new-onset diabetes at an advanced age. Pancreatic cancer often does not cause symptoms in the early stages. As cancer grows, it may start to cause symptoms. The symptoms may not be specific to pancreatic cancer, and they may come and go to begin with. This can make pancreatic cancer hard to diagnose.
Considering the high mortality rate and poor long-term survival of PDAC, medical experts have been developing various methods of diagnostics and treatment for this disease.
For PDAC patients with no metastasis, surgical resection is the ultimate goal to hopefully provide curative treatment. With the advancement of
endoscopic ultrasound (EUS) in both diagnosing and therapy for pancreatic cancer, it has provided treatment options not only by obtaining tissue samples to diagnose pancreatic cancer but also by providing more accurate local disease control while awaiting curative surgery and through palliative treatments for those with metastasis or advanced disease. The
Bumrungrad Digestive Disease Center team, led by US-trained
Clinical Associate Prof. Dr. Tossapol Kerdsirichairat, is one of the very few in the regions to implement endoscopic ultrasound-guided (EUS) and endoscopic retrograde pancreaticocholangiography (ERCP) interventions and regularly practice these techniques. Given that ERCP has become the standard of care worldwide to drain bile for those with obstructing bile tubes from PDAC, this article will only focus on the advances of interventional EUS for PDAC.
Obtaining quality samples of the mass helps to correctly diagnose cancer and its type, including genomic profiling, and provide a personalized treatment plan. EUS is a safe, effective, and efficient diagnostic tool in the evaluation of pancreatic mass lesions. As a diagnostic method for pancreatic cancer, EUS has proven rates of greater than 90%, especially for lesions less than 2-3 cm in size, in which it reaches a sensitivity rate of 99% compared to 55% for computer tomography (CT). Besides, EUS has a very high negative predictive value, which is critical to excluding small pancreatic lesions.
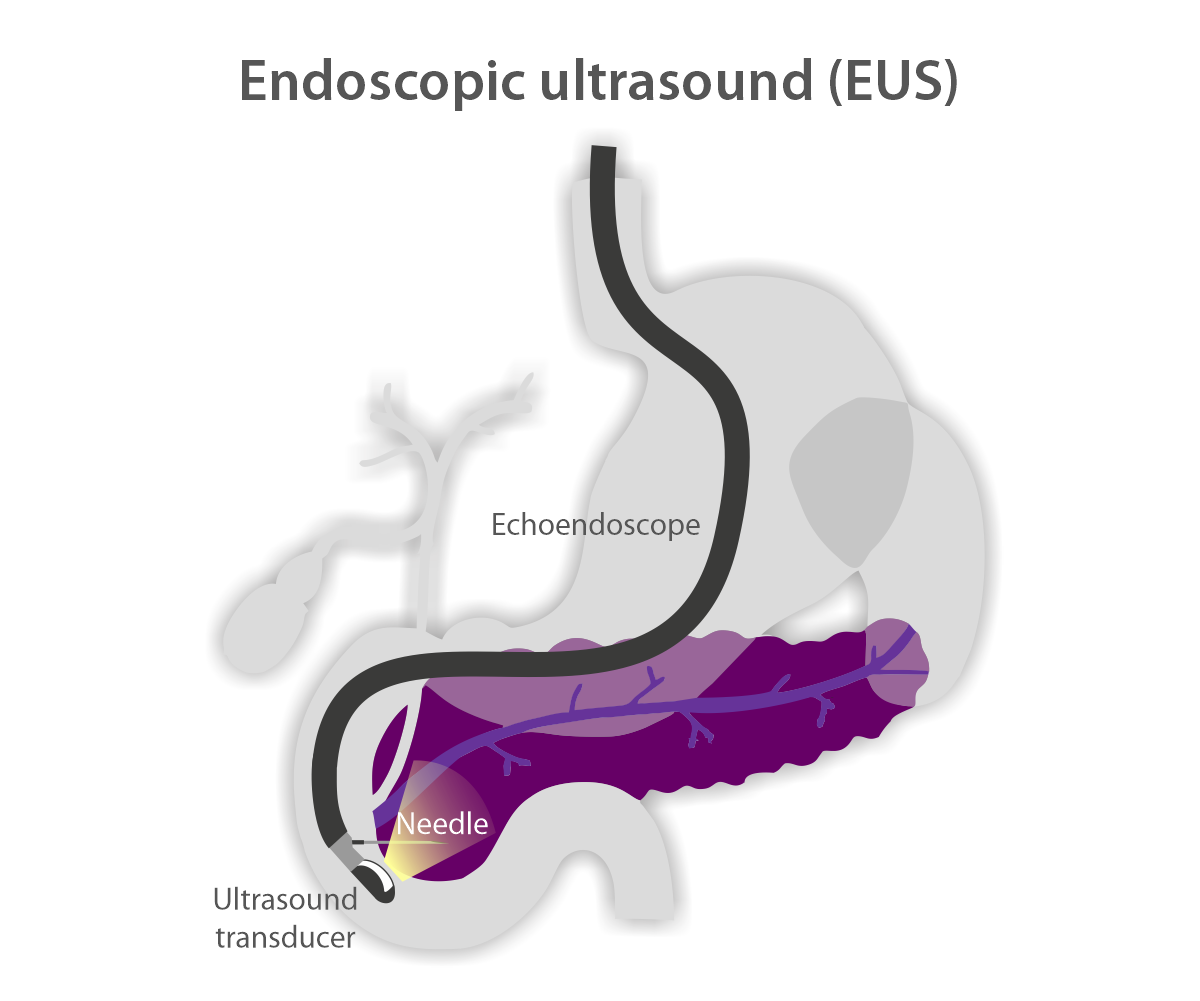
Endoscopic ultrasound (EUS) for pancreatic cancer management and treatment
EUS uses the same philosophy as a transabdominal ultrasound, which is widely used for pregnant women. While an obstetrician uses an ultrasound machine to visualize the fetus through the abdominal wall of the mother, an EUS is a compact ultrasound coupled with a finger-size tube-like endoscope with a camera at its tip, inserted through the mouth so that an endoscopist can visualize the head of the pancreas, which is located behind the small bowel wall, and the body and tail of the pancreas, which is located behind the wall of the stomach. The endoscope has a channel in its tube that an endoscopist can advance a device through, such as a needle, to obtain tissue, place a device, or inject medications. The safety of EUS is similar to that of transabdominal ultrasound-guided cord blood collection to collect the blood from the fetus, performed by an obstetrician. An endoscopist can use a real-time image to avoid blood vessels and vital structures.
- EUS-guided biliary drainage
Bile duct obstruction is common in pancreatic cancer patients, particularly on the right side of the pancreas, causing yellow discoloration of the eyes and skin, itchiness, and pale-colored stool. In general, bile duct obstruction from pancreatic cancer can be relieved by an endoscopic intervention called ERCP, for which an endoscopist can insert a small metal tube to expand the bile duct caliber against the pancreatic cancer that is pushing on the bile duct. Approximately 5–10% of patients may have complete occlusion at the level of the bile duct, small bowel, or stomach, precluding a standard ERCP. In this situation, an EUS-guided intervention to create a bypass between parts of the bile duct or the gallbladder and the stomach or the small bowel would be a salvage procedure. In selected patients, EUS-guided biliary drainage is more durable than percutaneous biliary drainage and is less invasive than surgical bypass.
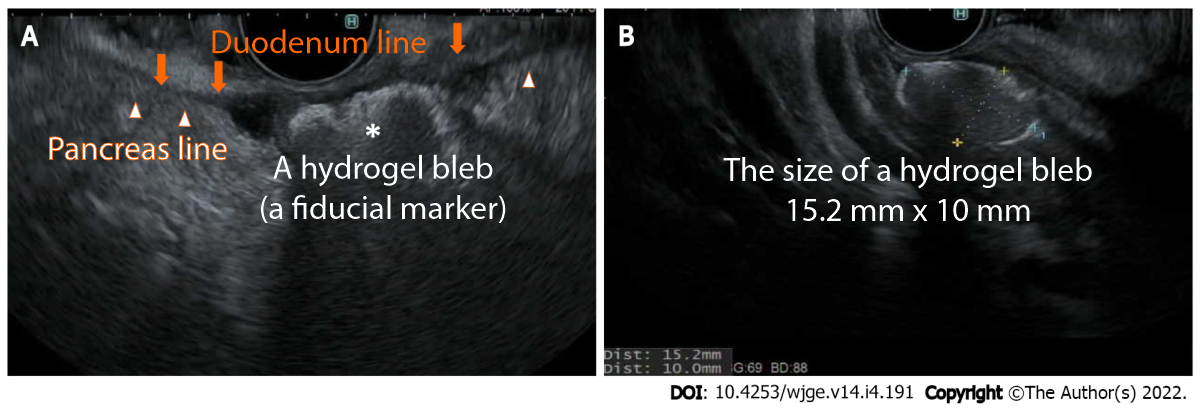
- EUS-guided placement of fiducial markers
In some patients with pancreatic cancer, surgery cannot completely remove all of the cancer cells. In this case, the combination of chemotherapy and/or radiation as the first step of treatment helps to shrink or stabilize the tumor before surgery. While chemotherapy can potentially control systemic disease, local disease could be controlled by radiation therapy. The use of fiducial markers (small metal objects or hydrogel) helps pinpoint the location of a tumor with greater accuracy at the time of radiation therapy, enhances the radiation effect on the tumor, and avoids possible injury to surrounding organs. Compared to percutaneously or surgically placed fiducials, EUS-guided fiducial placement is less invasive and may provide higher efficacy.
- EUS-guided celiac plexus neurolysis
A celiac plexus block is a pain relief treatment delivered by injection, which can be done by an EUS-guided approach as well. In patients with painful and inoperable pancreatic cancer, celiac plexus neurolysis considered early in the clinical course can improve pain and reduce morphine consumption.
- EUS-guided gastroenterostomy
Approximately 10–20% of patients with pancreatic cancer develop nausea and vomiting from obstruction at the level of the stomach and the small bowel from direct invasion of the cancer. EUS-guided gastroenterostomy creates a bypass for the gastric contents to enter the small bowel downstream beyond the obstruction point, with better and longer clinical success and fewer adverse events when compared to placing a metal tube in the small bowel to re-open the lumen. EUS-guided gastroenterostomy is less invasive when compared to the traditional surgical approach.
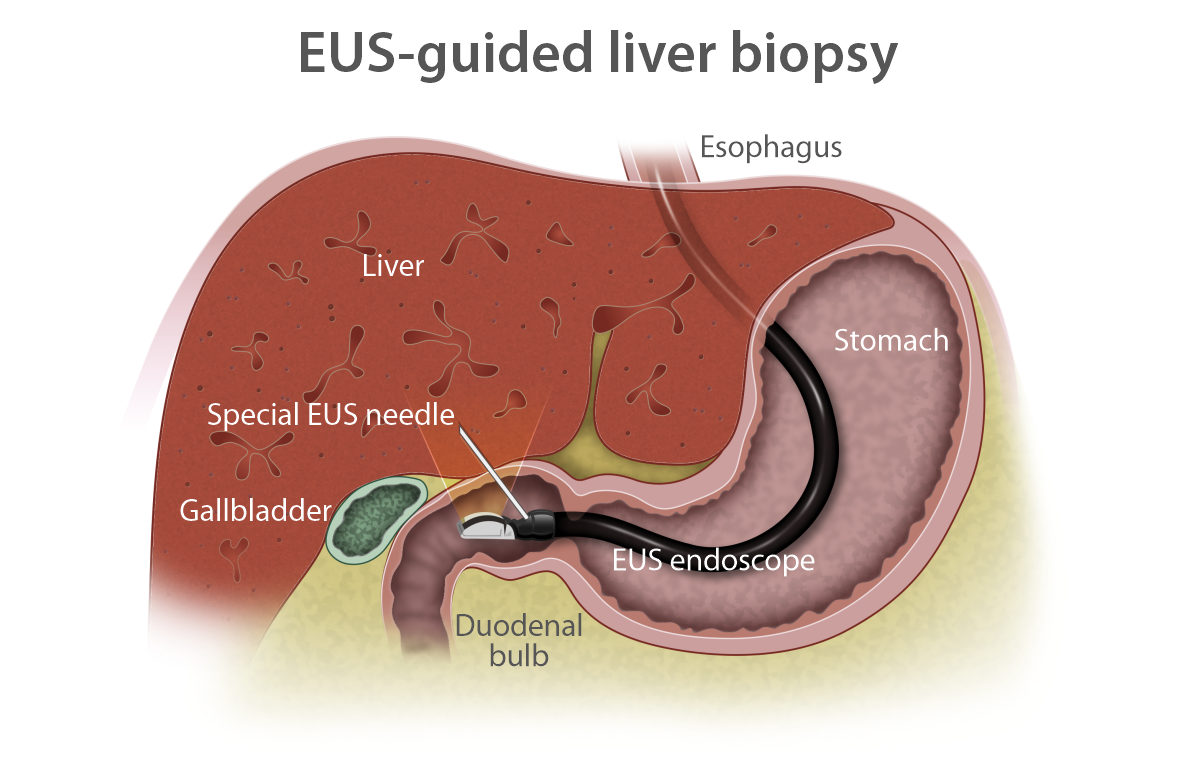
- EUS-guided liver biopsy
Some medications used to treat pancreatic cancer increase the risk of injury to the liver. In complicated or severe forms or for unclear causes of liver injury, liver biopsy can be used to confirm the cause of injury and/or to clarify the diagnosis. EUS-guided liver biopsy provides better tissue quality and improves the safety profile compared to percutaneous or through-blood-vessel approaches and is less invasive than surgical approaches. EUS-guided liver biopsy is preferred when other endoscopic procedures are indicated at the same time.
The studies of other EUS-guided interventions for pancreatic cancer listed below are still ongoing, with the hope of providing local disease control.
- EUS-guided brachytherapy
Brachytherapy is a form of radiation therapy used to treat various cancers by slowly releasing a small dose of radiation into the tumor over time. Treatment involves surgically placing radioactive seeds, capsules, or other implants directly into or near the cancerous tumor. In patients with unresectable pancreatic cancer, EUS can be used to place radioactive seeds into the pancreas, which is a deeply seated organ in the abdomen. This can result in partial remission of up to 80% in some groups of patients.
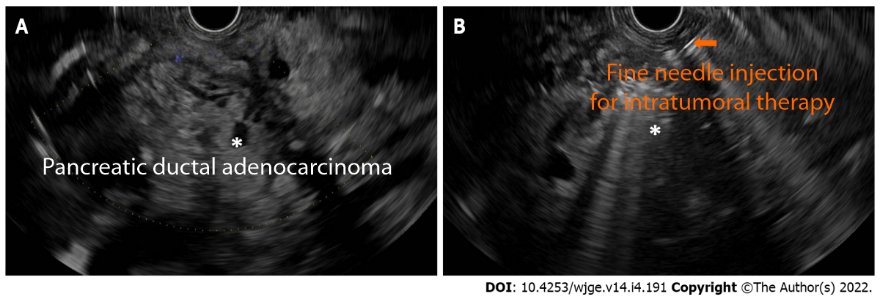
- EUS-guided injection of antitumoral agents
- Immunotherapy: Immunotherapeutic drugs are biologic agents used to enhance a patient's own immune system to fight cancer. In the setting of pancreatic cancer, these medications can be injected via a needle through an EUS-guided technique.
- Chemotherapy: Pancreatic cancer has a unique protective mechanism against chemotherapy. It forms a shell made of scar tissue as it grows. This shell precludes chemotherapy from getting into the cancer cells, resulting in a low rate of success. In selected groups of patients with unresectable pancreatic cancer, EUS-guided delivery of chemotherapeutic agents into the pancreatic mass could be considered.
- Gene therapy: EUS-guided injection might be useful in delivering gene therapy drugs directly to pancreatic cancer masses. The vast majority of clinical trials in this category are early in the process, including proof of concept and preclinical safety tests.
.png)
- EUS-guided ablative therapies
- Radiofrequency ablation is a local ablative method that can destroy the tumor by thermal coagulation and protein denaturation. In selected patients with an unresectable stage, EUS-guided radiofrequency ablation may improve survival benefits.
- Laser ablation uses a narrow and focused beam of light to cause injury to cancer cells. A laser probe can be advanced via the needle through an EUS-guided approach to provide local therapy in selected patients with unresectable pancreatic cancer.
- Photodynamic therapy has two steps: an injection of a photosensitizing agent, followed by the insertion of a needle into the targeted area to pass a small quartz optical fiber to illuminate and ablate tissue with the laser light. The expected degree of tumor damage could be measured to evaluate the treatment response. A phase 1 study in humans showed promising outcomes.
For more information about
endoscopic advances in the management of pancreatic cancer, please contact the Bumrungrad Digestive Disease Center to schedule an
appointment with our gastroenterologists.
For more information please contact:
Last modify: May 08, 2025